Efficacy
Proven efficacy in different patient profiles
Veltassa® allowed approximately 9/10 of patients to continue RAASi treatment, including an MRA.2–5
Proven efficacy with concomitant RAASi use backed by 4 placebo-controlled trials across co-morbidities2–5
The efficacy of Veltassa® has been evaluated across a variety of patient populations:
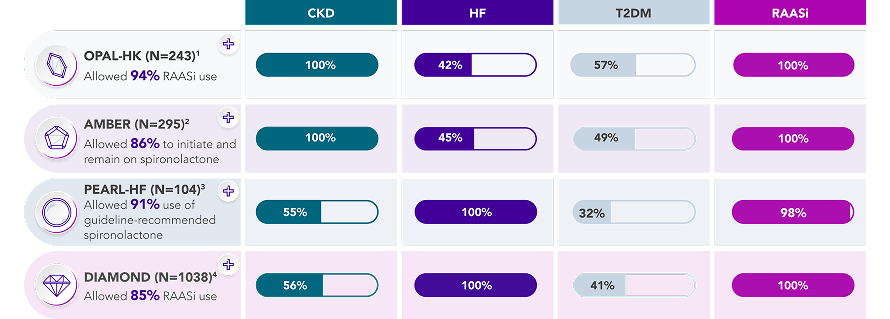
% bars represent the proportion of patients studied by baseline disease state across each clinical trial
RAASi therapy significantly reduces risk of progression to end-stage CKD6
In a separate study, mortality risk doubles when RAASi is reduced or stopped7
Proven effectiveness with HD patients in the real-world setting8,9
Veltassa® significantly reduced serum K+ in HD patients
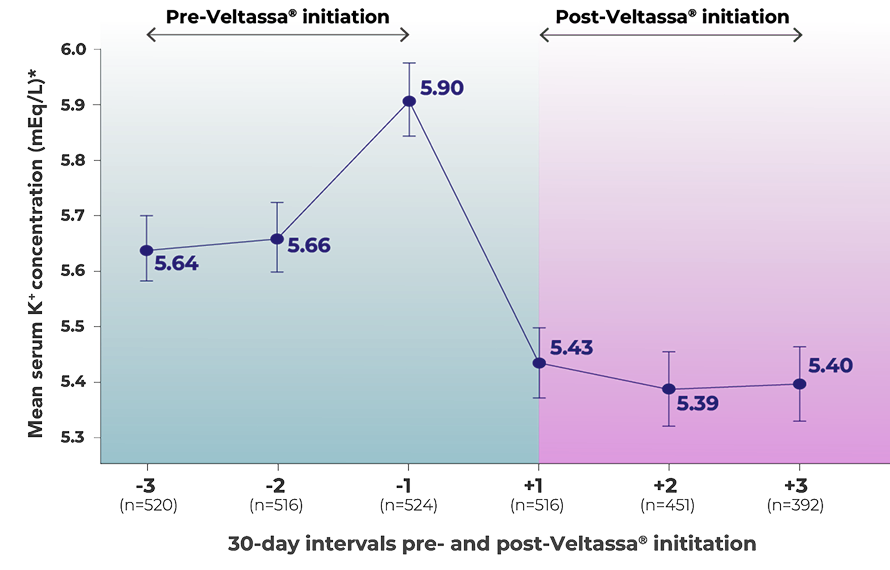
Adapted from Kovesdy et al. 2019.
Statistically significant reductions in K+, averaging approximately –0.5 mEq/L, were observed post-Veltassa® initiation (48% pre-Veltassa® vs 22% post-Veltassa® had K+ ≥6.0 mEq/L) (p<0.001)8
61% of patients initiated Veltassa® with 8.4 g once daily; this changed little over the study period.
A minority of patients (<20%) received a dose of 8.4 g but less frequently than once daily. Only 4% of patients initiated Veltassa® at the 16.8 g dosage.
DAVITA was a retrospective cohort study of 10,126 haemodialysis patients (527 in the Veltassa® cohort, 852 in the SPS cohort, and 8,747 in the non-K+ binder cohort), using electronic health data from DaVita Kidney Care in the United States.
Real-world data demonstrated that Veltassa®lowers and maintains serum K+ in ESRD patients undergoing HD9
Mean serum K+ before and after Veltassa® initiation2
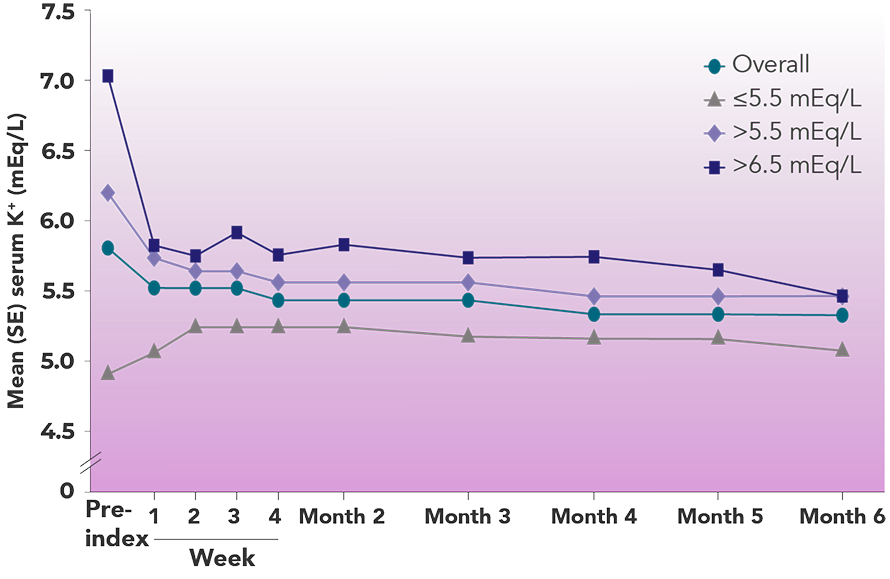
Adapted from Chatoth et al. 2017.
Overall, Veltassa® decreased serum K+ by 0.3 mEq/L by Week 1 and continued to decrease serum K+
Within the subgroups, serum K+ decreased most among those with higher pre-index serum K+ (>6.5 mEq/L subgroup decreased by 1.3 mEq/L by Week 1)8†
71.6% of patients were initially prescribed 8.4 g/d of Veltassa®. Mean daily Veltassa® dose during the post-index per iod: 7.9 g/d.
The FRENOVA study was a retrospective, descriptive cohort study of 268 haemodialysis patients at Fresenius Kidney Centre. Pre-index serum K+ subgroups ranged from ≤5.5 t o >6.5 mEq/L. 69.4% of patients initiated Veltassa® therapy on 8.4 g/day.
References & footnotes
Footnotes
*Using the last K+ value in each monthly interval for each included patient.
†Pre-index period was defined as up to 91 days prior to the start date on the first recorded Veltassa® prescription.
Abbreviations
CKD, chronic kidney disease; ESRD, end-stage renal disease; HD, haemodialysis; HF, heart failure; K+, potassium; MRA, mineralcorticoid receptor antagonist; RAASi, renin-angiotensin-aldosterone system inhibitor; T2DM, type 2 diabetes mellitus.
References
- Veltassa® European SmPC, 2024.
- Weir MR, et al. N Engl J Med. 2015;372(3):211–21.
- Agarwal R, et al. Lancet. 2019;394:1540–50.
- Pitt B, et al. Eur Heart J. 2011;32:820–8.
- Butler J, et al. Eur Heart J. 2022;43(41):4362–73.
- Brenner BM, et al. N Engl J Med. 2001;345:861–69.
- Epstein M, et al. Am J Manage Care. 2015;21:S212–20.
- Kovesdy CP, et al. Kidney Int Rep. 2019;4(2):301–9.
- Chatoth DK, et al. Outcomes in End-Stage Renal Disease Patients on Hemodialysis Taking Patiromer for Hyperkalemia. Presented at the American Society of Nephrology Kidney Week 2017; New Orleans, LA, November 2–5, Abstract TH-PO779.
HQ-PAT-2400064 | Date of preparation: February 2025
