Efficacy
Sustained K+ control
Sustained long term K+ control2
Mean serum K+ over 1 year in patients with CKD1,2*
AMETHYST DN trial
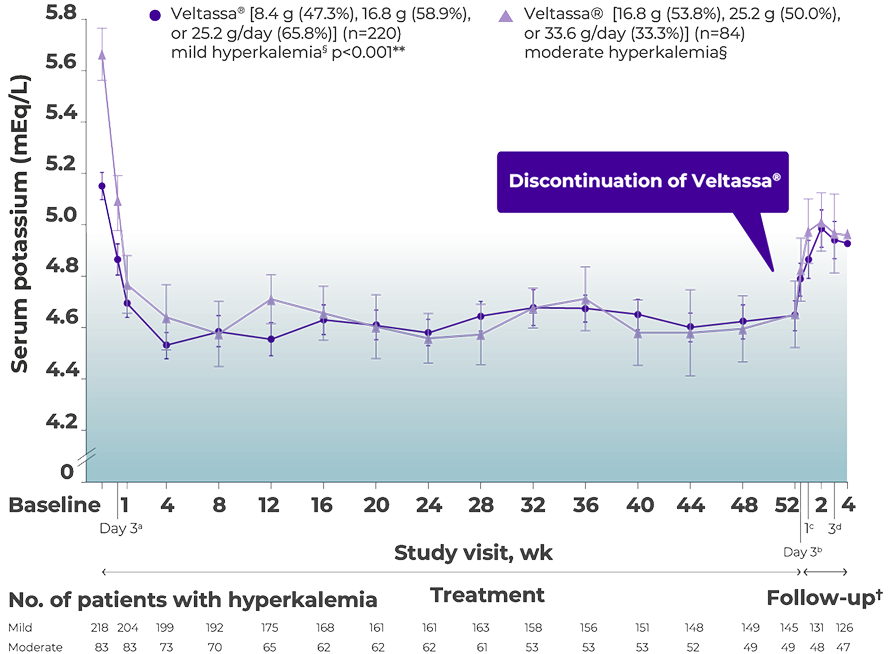
9/10
patients with moderate hyperkalemia sustained serum K+ within target range (3.8 to 5.0 mEq/L) over 1-year study period1,2*
a. At treatment Day 3, there were 202 patients with mild hyperkalemia and 82 with moderate hyperkalemia.
b. At follow-up Day 3, there were 163 patients with mild hyperkalemia and 58 with moderate hyperkalemia.
c. At follow-up Week 1, there were 154 patients with mild hyperkalemia and 57 with moderate hyperkalemia.
d. At follow-up Week 3, there were 126 patients with mild hyperkalemia and 48 with moderate hyperkalemia.
Veltassa® provides sustained long-term K+ control in HF patients3
Achieve long-term K+ control with Veltassa®:
Adjusted mean change in serum K+
DIAMOND TRIAL
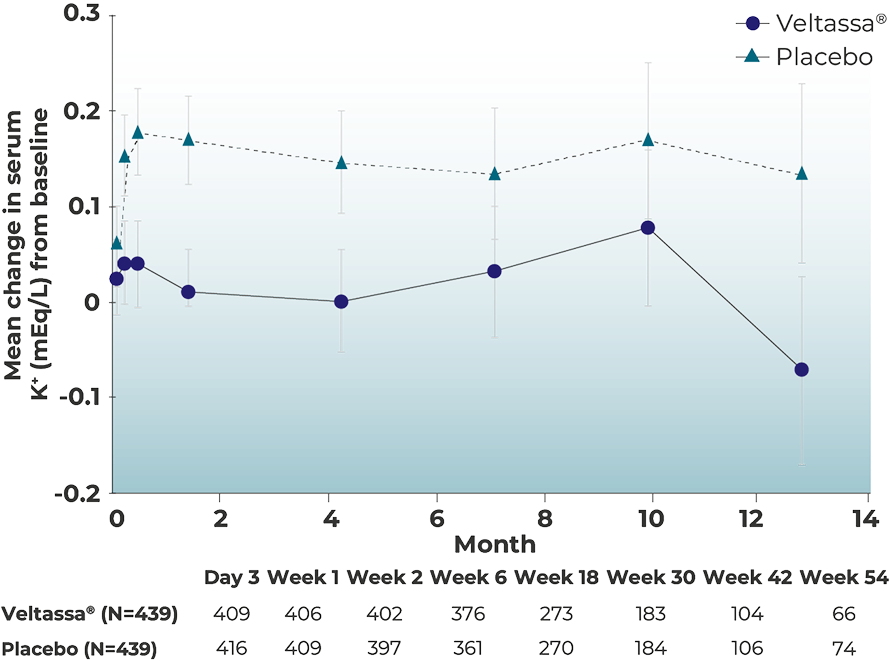
Adapted from Butler et al. 2022.
Patients were titrated up to a maximum of three packs/day; 8.4 g/pack in the run-in phase, patients who were randomised to Veltassa® continued the established number of packets of Veltassa®.
Randomised withdrawal phase
Patients on Veltassa® observed a between group-difference of –0.10 mEq/L vs placebo, in adjusted mean change in serum K+ levels (primary endpoint)
Veltassa® reduces healthcare resource utilisation and costs in patients with hyperkalemia4
In a 4-year retrospective US study, the cost and healthcare utilisation of hyperkalemia was evaluated in 3,078 patients with and without Veltassa®.
During the 6 month follow-up period, Veltassa® was associated with:

in total healthcare expenditures, despite 2x increase in outpatient pharmacy expenditure4
(p<0.05 vs no K+ binder; n=253 matched pairs)

in inpatient admissions or ED visits compared to patients who did not receive Veltassa®4
(p<0.05 vs no K+ binder; n=253 matched pairs)
References & footnotes
Footnotes
*Least squares mean (95% CI) serum potassium levels over 52 weeks and during post-treatment follow-up in patients with mild or moderate hyperkalemia (post-hoc mixed-effects models for repeated-measures analysis).2,3
†Increases in serum K+ may occur as early as 2 days after the last Veltassa® dose.1
§For adults, the recommended starting dose is 8.4 g patiromer once daily. The daily dose may be increased or decreased by 8.4 g, as necessary to reach the desired target range, up to a maximum dose of 25.2 g daily. For adolescents aged 12 to 17 years, the recommended starting dose is 4 g patiromer once daily. Adjust the daily dose of patiromer based on the serum potassium level and the desired target range, up to a maximum dose of 25.2 g daily.4
**At all points, p<0.001 (2-sided t-test) for least squares mean changes from baseline and Week 52 (or from last dose of patiromer received during the study).4
Abbreviations
CI, confidence interval; CKD, chronic kidney disease; ED, emergency department; HF, heart failure; HK, hyperkalemia; K+, potassium, RAASi, renin-angiotensin-aldosterone system inhibitor.
References
- Veltassa® European SmPC, 2024.
- Bakris GL, et al. JAMA. 2015;314(2):151–61.
- Butler J, et al. Eur Heart J. 2022;43(41):4362–73.
- Desai NR, et al. J Med Econ. 2021;24(1):1025–36.
HQ-PAT-2400063 | Date of preparation: February 2025
