Efficacy
Works from the 1st dose
Effective from the first dose, regardless of HK severity2
Greatest K+ reduction observed in patients with severe hyperkalemia2
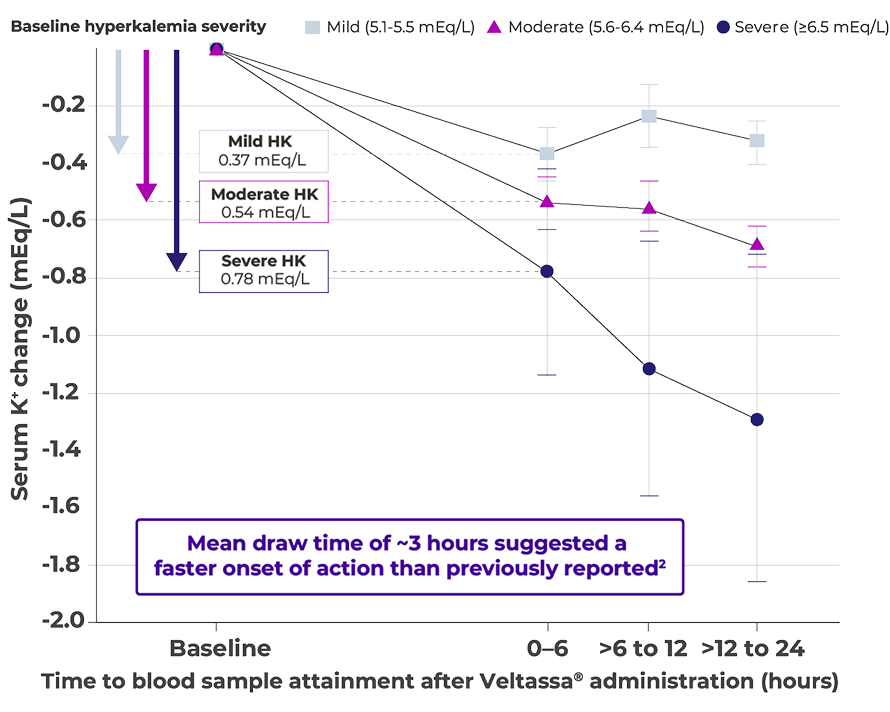
Adapted from Di Palo et al. 2022.

Significant reduction in K+
Significant mean reduction in serum K+ levels of 0.50 mEq/L from baseline (P<0.001; primary endpoint), within 6 hours after Veltassa® was administered
Real-world retrospective cohort study of 881 patient encounters for acute, non-life threatening hyperkalemia treated in emergency departments (n=152), inpatient units (n=679), and intensive care units (n=50) at the Montefiore Medical Centre. Mean baseline serum K+ levels of 5.60 mEq/L. The lowest dose of Veltassa® was 8.4 g used in 721 encounters (81.8%), 16.8 g was used in 154 encounters (17.5%), and 25.2 g was used in 6 encounters (0.7%). In the 24 hours after the initial administration of Veltassa®, in 725 encounters (82.3%), patients received no further doses of K+ binders, whereas in 137 encounters (15.5%), patients received 1 additional dose and in 19 encounters (2.2%), patients received 2 or more additional doses.
Consistent K+ reduction over 48 hours3
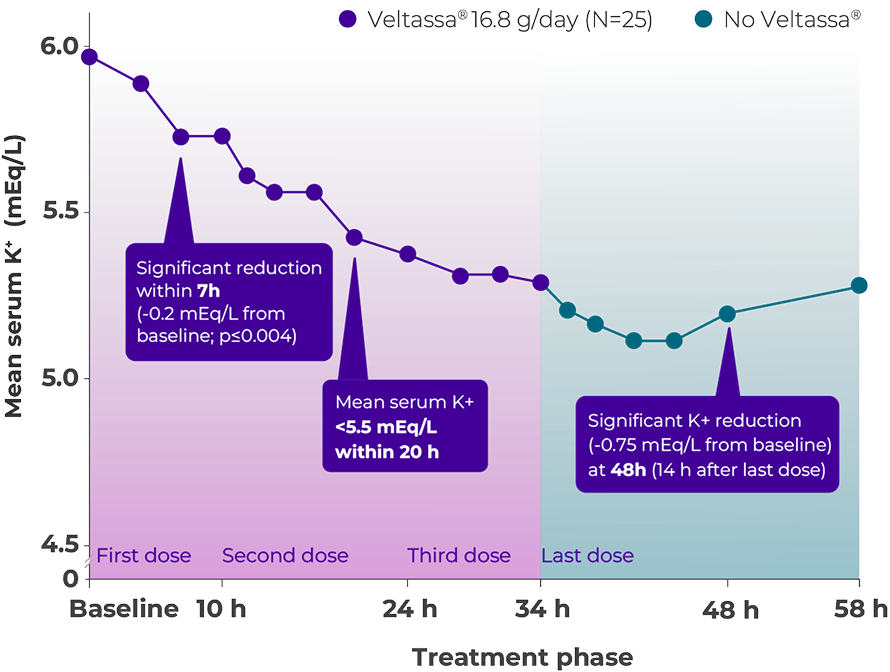
Adapted from Bushinsky et al. 2015.
Phase 1, open-label, single-arm study. 25 patients [moderate-to-severe hyperkalemic CKD patients receiving at least one RAASi while consuming a 60 mEq/day potassium diet] with baseline serum K+ levels of 5.93 mEq/L entered a 2-day run-in phase before receiving Veltassa® 8.4 g/ dose with morning and evening meals for a total of four doses.
References & footnotes
Abbreviations
CKD, chronic kidney disease; h, hour; HK, hyperkalemia; K+, potassium; RAASi, renin-angiotensin-aldosterone system inhibitor.
References
- Veltassa® European SmPC, 2024.
- Di Palo KE, et al. JAMA Netw Open 2022;5(1):e2145236.
- Bushinsky DA et al. Kidney Int. 2015;88(6):1427–33.
HQ-PAT-2400062 | Date of preparation: February 2025
